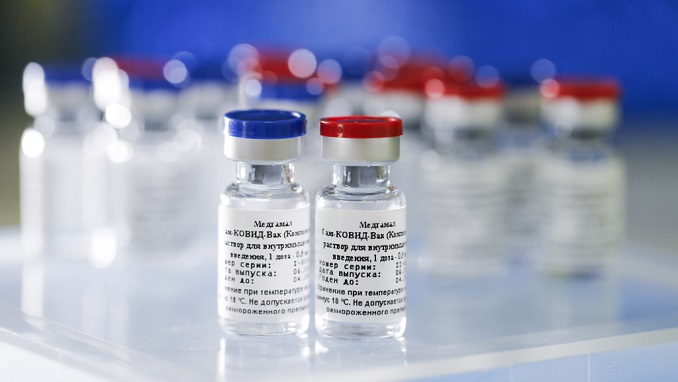
American doctors and medical workers took part in the first briefing on the Russian coronavirus vaccine in the United States and expressed strong interest in Sputnik V, the Russian Direct Investment Fund (RDIF) said, Sputnik News has reported.
“More than one hundred doctors and medical workers from Maimonides Medical Center, one of the leading U.S. hospitals at the forefront of the fight against the COVID-19 pandemic since March 2020, expressed strong interest in Russia’s Sputnik V vaccine at an online briefing, the first of its kind, with Russian scientists from the Gamaleya National Research Center of Epidemiology and Microbiology,” the sovereign wealth fund said in a press release.
Scientists from the Gamaleya Center told the U.S. audience about the development of the Russian vaccine and the progress of the ongoing post-registration trial.
The Russian researchers put an emphasis on the advantages of the human adenoviral vector platform used in Sputnik V over untested technologies such as the monkey adenoviral vector platform or mRNA technology, which have not been studied for potential side effects such as impact on fertility or cancer risks over the long term.
Gamaleya’s Unique Technology
They also shared information about the Gamaleya Centre’s own unique technology with the American doctors, which is a two-shot model using two different human adenoviral vectors, Ad5 and Ad26. This approach makes Sputnik V, the world’s first coronavirus vaccine, different from other potential vaccines based on human adenoviral vectors using only one type of adenoviral vector in a single-shot model.
The U.S. doctors were “very excited” to hear about the vaccine, said Dr Jacob Shani, Chair of the Heart and Vascular Center and the Department of Cardiology at Maimonides Medical Center.
“We read the publication in The Lancet and this publication was also very impressive. It showed good results of the Phase 1 and 2 trials, including the development of antibodies, cellular immunity, and only minimal side effects. It was all very encouraging. On a personal level we were impressed with the Gamaleya scientists. They were very professional, knowledgeable, and most importantly, they appeared to be very transparent. They answered every question without hesitation,” Shani said, as quoted in the press release.
The key mission of the Russian scientists is to provide “timely and detailed information about the Sputnik V vaccine in a transparent manner for all interested parties,” said Dr Alexander Gintsburg, Head of the Gamaleya National Research Centre of Epidemiology and Microbiology.
“We were delighted to share this information with colleagues from the Maimonides Medical Centre, one of the most respected U.S. medical institutions directly involved in fighting the COVID-19 pandemic. We also appreciated the professional, non-partisan approach by the U.S. doctors, who, just as their Russian colleagues, are tired of the pandemic and would like to see a working, effective, and safe vaccine against COVID-19 as soon as possible. We regard international cooperation on the vaccine as key to a decisive victory over COVID-19 and will work towards this goal with our partner, the Russian Direct Investment Fund,” he said.
World’s First COVID-19 Vaccine
Russia registered the world’s first coronavirus vaccine, named Sputnik V and developed by the Gamaleya Center, on 11 August. The vaccine was registered after two successful phases of testing proved that it was capable of building immunity against the virus in all of the volunteers involved in the tests. Currently, Russia is carrying out post-registration clinical trials of the drug, involving over 42,000 people in Russia and around the world.
The CEO of the Russian Direct Investment Fund, Kirill Dmitriev, previously revealed that the fund had received requests from 27 countries for Sputnik V.